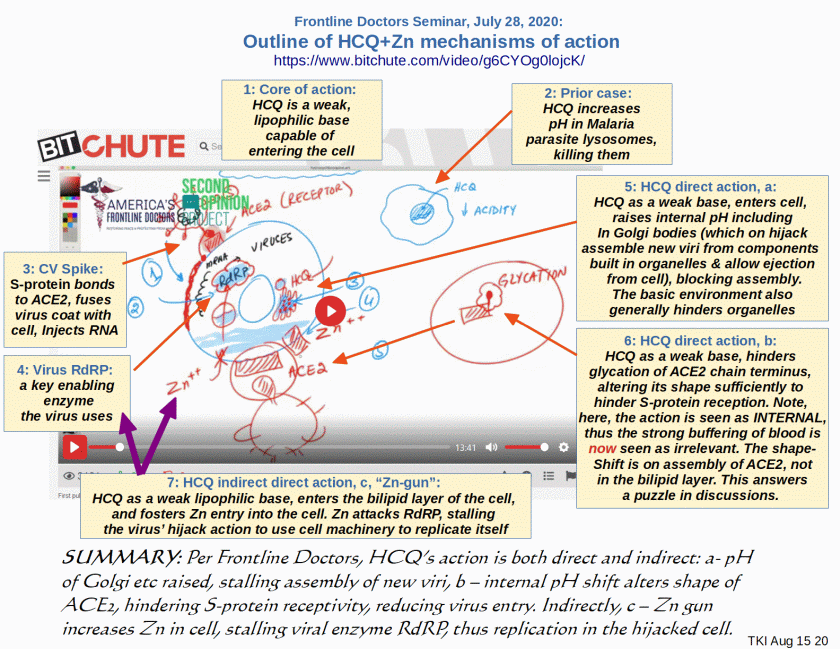
In their July 28 seminar, the Frontline Doctors Group led by Dr Simone Gold, have put some plausible mechanisms for HCQ based cocktails on the table. These were noted on in an augmentation to an earlier post, but deserve headlining in their own right:
Dr Frieden OP: >>I have found at Bit Chute, a July 28 Frontline Doctors seminar which describes several mechanisms of action. Accordingly, I take liberty to annotate a screenshot, summarising several mechanisms of action described by these Doctors [cf. here for their references], but which are hard to find because of now almost pervasive censorship:
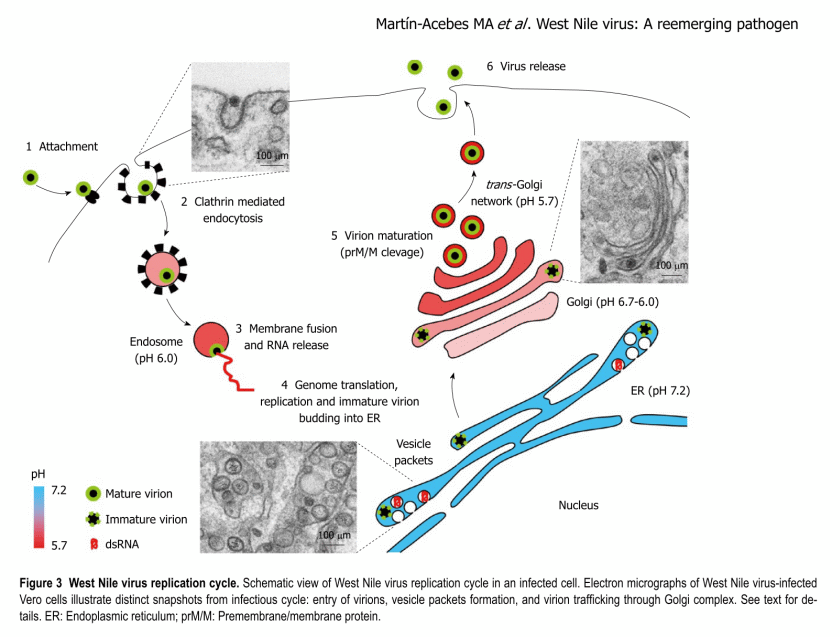
I add, that the above suggests a fairly similar viral attack process to the West Nile Virus (which is also an RNA virus), e.g.:
U/D, Mon Aug 16: I further add, a “DrBeen” — Dr Mobeen Syed — educational video, just found — this is the presenter summarised above:
I note, this first answers a puzzle on the mode of action, shape-shift of ACE2: the shift is INTERNAL to the cell by hindering “glycation” of the final AA (thus prior to exposure to buffering of blood etc), altering the shape enough to hamper S-protein reception. This reduces fusion with bilipid layer and RNA injection.
Other direct mechanisms as noted, reduce intracellular acidity thus action of organelles. They highlight stalling of assembly of new viri in the Golgi bodies, with implication of blocking export of fresh viri, thus hampering the multiplication chain. The by now well known indirect activity is that as a lipophilic molecule, HCQ enters the cell bilipid layer membrane, acting as a Zn ionophore, i.e. it “shoots” Zn into the cell. Zn in turn hinders a key viral enzyme, RdRP.
Thus, we see a plausible picture of causal action, involving multiple, synergistic effects. This lends credibility to the use of HCQ-based cosctails in treating the early viral phases of CV19.>>
Unfortunately, WP for UD is not set up to embed BitChute videos.
An odd bit of support for this, is that HCQ/CQ have been used as fish tank cleaner for about 40 years. The complex animals (the fish) live, but the crud from several kingdoms, dies. That points to attack modes that hit core cell processes, such as we may summarise:
That seems to be what is now on the table, through the effects of pH shifting, as proposed. In short, we have reasonable mechanisms to go with the reports of doctors who are treating CV 19 in the early, viral phase, with vulnerable group patients:
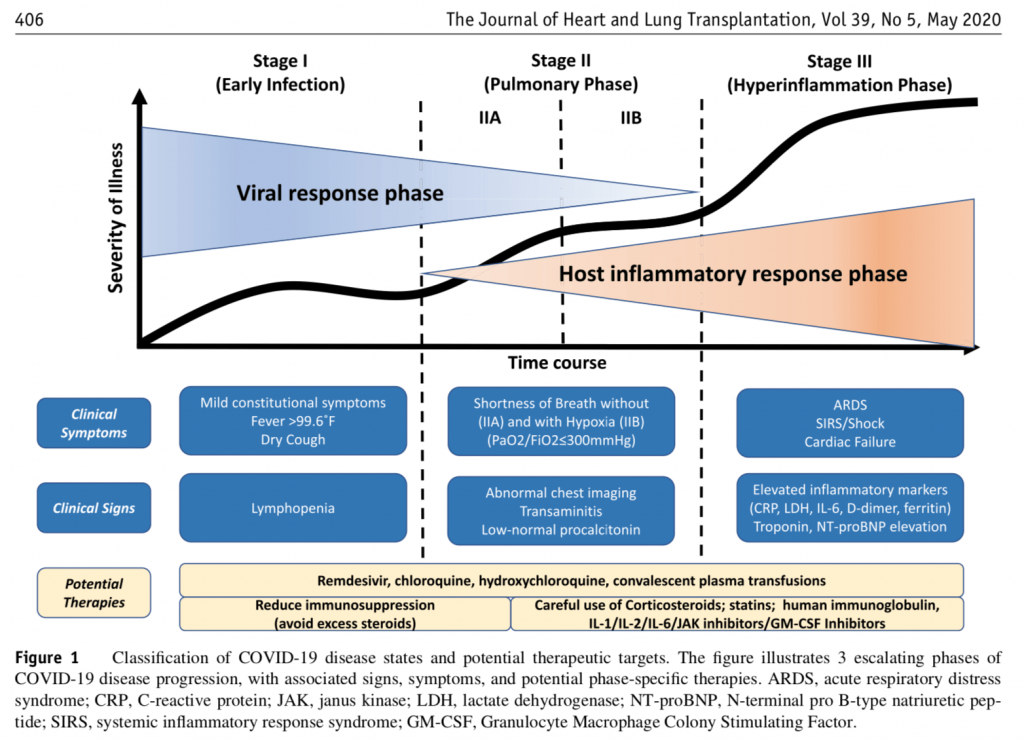
From this, we can freely say that it is going to be a challenge to refute the framework of issues and implicit model being presented in the open letter to Dr Fauci:
>>There is currently no recommended pharmacologic early outpatient treatment for individuals in the flu stage of the illness, correct?
It is true that COVID-19 is much more lethal than the flu for high-risk individuals such as older patients and those with significant comorbidities, correct?
Individuals with signs of early COVID-19 infection typically have a runny nose, fever, cough, shortness of breath, loss of smell, etc., and physicians send them home to rest, eat chicken soup etc., but offer no specific, targeted medications, correct?
These high-risk individuals are at high risk of death, on the order of 15% or higher, correct?
So just so we are clear—the current standard of care now is to send clinically stable symptomatic patients home, “with a wait and see” approach?
Are you aware that physicians are successfully using Hydroxychloroquine combined with Zinc and Azithromycin as a “cocktail” for early outpatient treatment of symptomatic, high-risk, individuals?
Have you heard of the “Zelenko Protocol,” for treating high-risk patients with COVID 19 as an outpatient?
Have you read Dr. Risch’s article in the American Journal of Epidemiology of the early outpatient treatment of COVID-19?
Are you aware that physicians using the medication combination or “cocktail” recommend use within the first 5 to 7 days of the onset of symptoms, before the illness impacts the lungs, or cytokine storm evolves?
Again, to be clear, your recommendation is no pharmacologic treatment as an outpatient for the flu—like symptoms in patients that are stable, regardless of their risk factors, correct?
Would you advocate for early pharmacologic outpatient treatment of symptomatic COVID-19 patients if you were confident that it was beneficial?
Are you aware that there are hundreds of physicians in the United States and thousands across the globe who have had dramatic success treating high-risk individuals as outpatients with this “cocktail?”
Are you aware that there are at least 10 studies demonstrating the efficacy of early outpatient treatment with the Hydroxychloroquine cocktail for high-risk patients — so this is beyond anecdotal, correct?
If one of your loved ones had diabetes or asthma, or any potentially complicating comorbidity, and tested positive for COVID-19, would you recommend “wait and see how they do” and go to the hospital if symptoms progress?
Even with multiple studies documenting remarkable outpatient efficacy and safety of the Hydroxychloroquine “cocktail,” you believe the risks of the medication combination outweigh the benefits?
Is it true that with regard to Hydroxychloroquine and treatment of COVID-19 infection, you have said repeatedly that “The Overwhelming Evidence of Properly Conducted Randomized Clinical Trials Indicate No Therapeutic Efficacy of Hydroxychloroquine (HCQ)?”
But NONE of the randomized controlled trials to which you refer were done in the first 5 to 7 days after the onset of symptoms- correct?
All of the randomized controlled trials to which you refer were done on hospitalized patients, correct?
Hospitalized patients are typically sicker that outpatients, correct?
None of the randomized controlled trials to which you refer used the full cocktail consisting of Hydroxychloroquine, Zinc, and Azithromycin, correct?
While the University of Minnesota study is referred to as disproving the cocktail, the meds were not given within the first 5 to 7 days of illness, the test group was not high risk (death rates were 3%), and no zinc was given, correct?
Again, for clarity, the trials upon which you base your opinion regarding the efficacy of Hydroxychloroquine, assessed neither the full cocktail (to include Zinc + Azithromycin or doxycycline) nor administered treatment within the first 5 to 7 days of symptoms, nor focused on the high-risk group, correct?
Therefore, you have no basis to conclude that the Hydroxychloroquine cocktail when used early in the outpatient setting, within the first 5 to 7 days of symptoms, in high risk patients, is not effective, correct?
It is thus false and misleading to say that the effective and safe use of Hydroxychloroquine, Zinc, and Azithromycin has been “debunked,” correct? How could it be “debunked” if there is not a single study that contradicts its use?
Should it not be an absolute priority for the NIH and CDC to look at ways to treat Americans with symptomatic COVID-19 infections early to prevent disease progression?
The SARS-CoV-2/COVID-19 virus is an RNA virus. It is well-established that Zinc interferes with RNA viral replication, correct?
Moreover, is it not true that hydroxychloroquine facilitates the entry of zinc into the cell, is a “ionophore,” correct?
Isn’t also it true that Azithromycin has established anti-viral properties?
Are you aware of the paper from Baylor by Dr. McCullough et. al. describing established mechanisms by which the components of the “HCQ cocktail” exert anti-viral effects?
So- the use of hydroxychloroquine, azithromycin (or doxycycline) and zinc, the “HCQ cocktail,” is based on science, correct?>>
Let us see how the “game” moves forward beyond this point. END
PS: As an extra, here is Dr Zelenko:
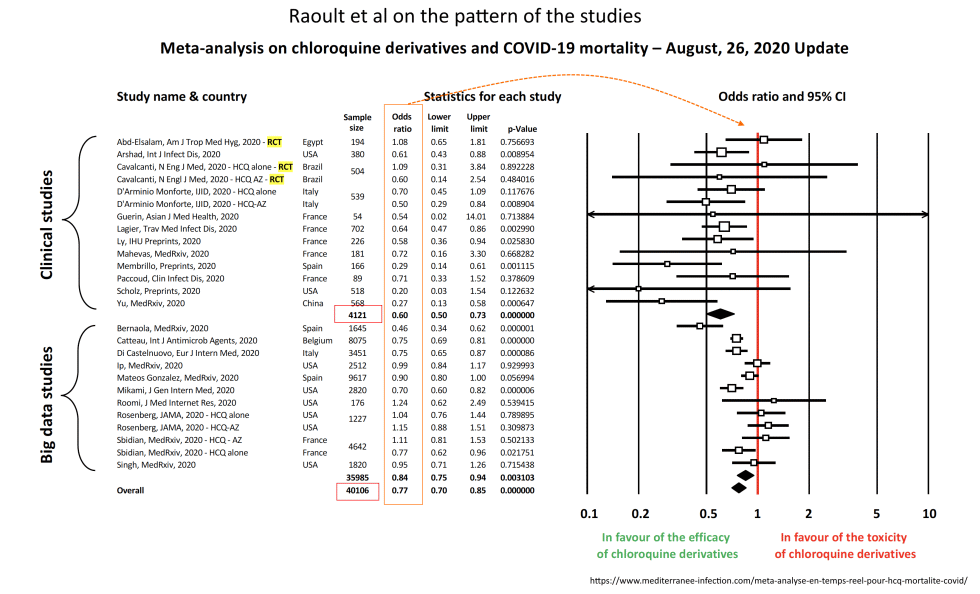
PPS: As a further extra, Aug 29, HT Jerry, Raoult et al on the pattern of cases and studies, involving 40,000+ cases: