Louise Lerner writes:
For the past two centuries, humans have relied on fossil fuels for concentrated energy; hundreds of millions of years of photosynthesis packed into a convenient, energy-dense substance. But that supply is finite, and fossil fuel consumption has tremendous negative impact on Earth’s climate.
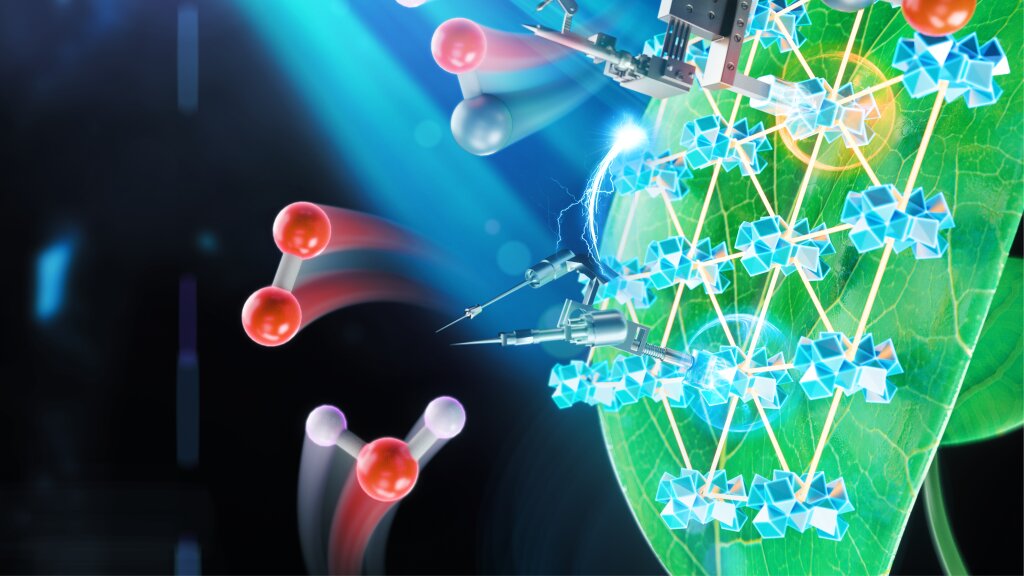
“The biggest challenge many people don’t realize is that even nature has no solution for the amount of energy we use,” said University of Chicago chemist Wenbin Lin. Not even photosynthesis is that good, he said: “We will have to do better than nature, and that’s scary.”
One possible option scientists are exploring is “artificial photosynthesis“—reworking a plant’s system to make our own kinds of fuels. However, the chemical equipment in a single leaf is incredibly complex, and not so easy to turn to our own purposes.
A Nature Catalysis study from six chemists at the University of Chicago shows an innovative new system for artificial photosynthesis that is more productive than previous artificial systems by an order of magnitude. Unlike regular photosynthesis, which produces carbohydrates from carbon dioxide and water, artificial photosynthesis could produce ethanol, methane, or other fuels.
Though it has a long way to go before it can become a way for you to fuel your car every day, the method gives scientists a new direction to explore—and may be useful in the shorter term for production of other chemicals.
“This is a huge improvement on existing systems, but just as importantly, we were able to lay out a very clear understanding of how this artificial system works at the molecular level, which has not been accomplished before,” said Lin, who is the James Franck Professor of Chemistry at the University of Chicago and senior author of the study.
‘We will need something else’
“Without natural photosynthesis, we would not be here. It made the oxygen we breathe on Earth and it makes the food we eat,” said Lin. “But it will never be efficient enough to supply fuel for us to drive cars; so we will need something else.”
The trouble is that photosynthesis is built to create carbohydrates, which are great for fueling us, but not our cars, which need much more concentrated energy. So researchers looking to create alternates to fossil fuels have to re-engineer the process to create more energy-dense fuels, such as ethanol or methane.
In nature, photosynthesis is performed by several very complex assemblies of proteins and pigments. They take in water and carbon dioxide, break the molecules apart, and rearrange the atoms to make carbohydrates—a long string of hydrogen-oxygen-carbon compounds. Scientists, however, need to rework the reactions to instead produce a different arrangement with just hydrogen surrounding a juicy carbon core—CH4, also known as methane.
This re-engineering is much trickier than it sounds; people have been tinkering with it for decades, trying to get closer to the efficiency of nature.
Even with the significantly improved performance, however, artificial photosynthesis has a long way to go before it can produce enough fuel to be relevant for widespread use. “Where we are now, it would need to scale up by many orders of magnitude to make an sufficient amount of methane for our consumption,” Lin said.
Complete article at Phys.org.
Perhaps the reason that “the efficiency of nature” is so far beyond our ability to duplicate is that living systems are unnatural, in the sense that they were not made by natural processes.