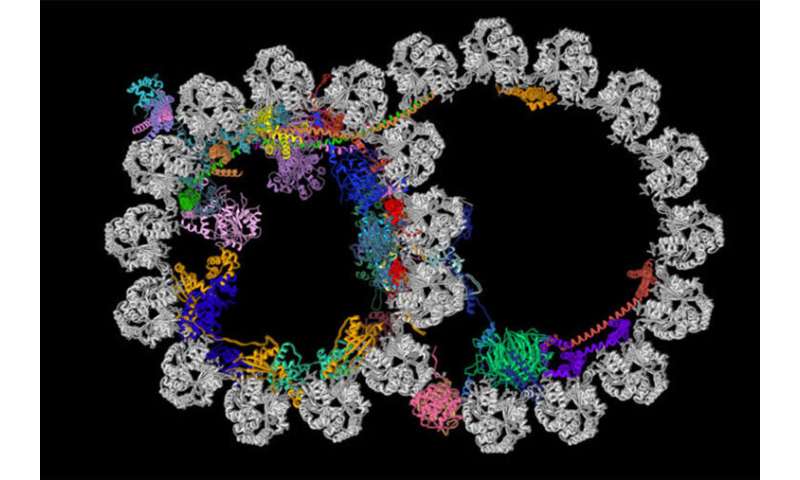
Cilia contain structures called ciliary doublet microtubules. Pictured is a cross section of one of these structures. A new study from the School of Medicine and Harvard Medical School has described the most detailed picture yet of these vital cellular structures. The microtubule is shown in gray, and the newly identified proteins decorating the microtubule are depicted in various colors. Credit: R. Zhang, A. Brown
Cilia (or flagella)—whiplike appendages on cells—do a lot of jobs that keep the body healthy:
“Before this work, everyone assumed these proteins inside cilia just stabilize the structure, which is true for a subset of the proteins, especially when you consider the forces produced by the continuous beating of the cilia,” Zhang said. “But based on how they are arranged inside this structure, we believe these proteins are doing many more things.”
Since many of the proteins protrude through the cilia, Zhang and his colleagues speculate that they may allow for communication between the inside and the outside of the ciliary microtubule doublets; govern the function of enzymes that make important biochemical reactions possible; and sense changes in the calcium concentration of the environment, which plays a role in triggering the cilia to beat.
Julia Evangelou Strait, “Scientists unravel mysteries of cells’ whiplike extensions” at Phys.Org
Follow UD News at Twitter!
: