A team of researchers led by Professor Sijbren Otto of the University of Groningen, in the Netherlands, has announced that it has observed not only self-replication, but also mutants and even new “species,” in a bunch of molecules in the lab. Does this research show how life might have arisen spontaneously, or is it nothing more than a case of intelligent design by clever chemists?
In today’s post, I’m going to argue that the claims made by Professor Otto and his team are flawed, on no less than seven counts. But before I examine their press release and their paper in Nature Chemistry, I’d like to discuss a Science LinX video that was posted on Youtube last year (March 17, 2015), titled, “Chemical evolution: creating life?”, which explains the work being carried out Otto and his group:
The text of the video reads as follows (note: all bold emphases shown in this post are mine – VJT):
This is Sijbren Otto, a chemistry researcher from Groningen. A while ago, he and his research group discovered molecules that can reproduce. Start out with a handful, and after a while, you’ve got twice as many. A bit later, there are four times as many, and so on. There are even different kinds of reproducing molecules that compete for building blocks. “That looks exactly like animals competing for food,” Otto thought. Now, he wants to trick molecules into real evolution – you know, the Charles Darwin kind of evolution that all living organisms have been going through for some four billion years. Who knows? His research might one day result in some kind of chemical life form.
It all started with a pot of pretty simple molecules able to couple on two sides. Also, they have a little tail that exactly fits the tail of the other molecules. The idea was that this would help create some structure. And so it did. The molecules hook up and form rings, counting six, seven or eight molecules each. And as the tails fit onto each other, so do the rings. A six-ring fits a six-ring, a seven-ring fits a seven-ring, and so on. Stacks of rings become towers. Towers become long threads. If you stir the pot, the threads will break apart. But each piece will start to grow again – from both ends. Soon, Otto’s pots were teeming with small threads of molecules.
However, in real evolution, species also change when they reproduce every once in a while. A mutation causes a child to be slightly different from its parents. So, Otto’s students started working with several different tails that might sometimes not fit together so well. When the molecules stack, mistakes happen. These errors may cause the thread to grow faster, or to stop growing, or to grow into complicated shapes, or to do something else yet. In fact, Otto doesn’t know, because this is what his students are finding out now. In any case, Otto’s pots are brewing. The question is: when can it be considered life? Otto thinks there is a gray area between “really alive” and “really dead.” But when it waves at us and says, “Hello,” you can be pretty sure it’s alive.
The latest press release: “The origin of abiotic species”
On January 4, 2016, a press release titled, The origins of abiotic species, was put out by the University of Groningen. The key part reads as follows:
Otto has been working on chemical evolution for several years now. ‘It started with a chance discovery’, he explains. ‘We found some small peptides that could arrange themselves into rings, which could then form stacks.’ Once a stack began to form, it would continue to grow and would then multiply by breaking into two smaller stacks. These would both grow and break again, and so on. The stacks also stimulated the formation of the rings from which they are composed. The stacks and rings are called ‘replicators‘, as they are able to make copies of themselves.
Jan Sadownik, a postdoc in the Otto group, discovered that if you offer the replicators two different types (A and B) of building blocks (‘food’) they will make copies of themselves. He observed the emergence of a set of replicator mutants that specialized in food A, but also incorporated some B. The rings mainly comprised the A building blocks, with just a few B’s.
Some days later Sadownik saw a second set of mutants emerge that specialized in food B, but also tolerated some A. This second set proved to be a descendant of the first set, which meant there was an ancestral relationship between the sets. This is very similar to how new species form from existing ones during biological evolution, except that this process of species formation does not involve full-fledged biological organisms, but occurs instead at the molecular level.
Molecular speciation
Looking at the molecular ‘speciation’ process in more detail allowed the researchers to identify specific mutants within the first set of replicators that were responsible for the generation of the second set. They had therefore established the mechanism by which replicator ‘species’ form with unprecedented molecular-level detail….
This shows how new ‘species’ can emerge through chemical evolution. Otto explains, ‘Of course, the term speciation should only be used when referring to sexually reproducing organisms, but our work shows much the same patterns.‘ The exciting part, says Otto, is that ‘we start with no replicators, but see first one type emerge and then after a while, another. That is certainly most significant!’
One physicist’s take on the new research
Physicist Rob Sheldon made a pithy observation regarding the University of Groningen’s latest press release and the new paper by Sandownik, Mattia, Nowak and Otto: “If this paper were about anything but ‘peptides’, it would be called ‘crystallization’.”

|
Indeed. Crystals have been shown to exhibit many of the properties displayed by Professor Otto’s peptide structures. I would invite skeptical readers to have a look at a 2013 article titled, It’s almost alive! Scientists create a near-living crystal by Brandon Keim (Wired, January 31, 2013). Written in a tone of breathless anticipation, the article exudes the same kind of starry-eyed optimism as the University of Groningen press release, as it reports on how particles of a mineral called hematite (pictured above, image courtesy of Wikipedia) can be coaxed into forming “living crystals”:
Three billion years after inanimate chemistry first became animate life, a newly synthesized laboratory compound is behaving in uncannily lifelike ways.
The particles aren’t truly alive — but they’re not far off, either. Exposed to light and fed by chemicals, they form crystals that move, break apart and form again.
“There is a blurry frontier between active and alive,” said biophysicist Jérémie Palacci of New York University….
Palacci and fellow NYU physicist Paul Chaikin led a group of researchers in developing the particles, which are described Jan. 31 in Science as forming “living crystals” in the right chemical conditions….
Chaikin notes that life is difficult to define, but can be said to possess metabolism, mobility, and the ability to self-replicate. His crystals have the first two, but not the last.…
As for what’s happening now in Palacci and Chaikin’s lab, a particle currently under development isn’t mobile, but it has a metabolism and is self-replicating.
“We’re working on it,” Chaikin said.
The abstract of Palacci and Chaikin’s paper can be found here.
Now, to be fair, I should point out that the structures created by the University of Groningen team are capable of a kind of self-replication – that is, if you’re willing to call breaking in two “replication.” But that’s about the only thing they can do, that Palacci and Chaikin couldn’t – and in any case, they state towards the end of the article that they have now created a self-replicating particle which has a metabolism. So on the whole, I think Rob Sheldon’s remarks are pretty accurate. These peptide structures are no more alive than some crystals are.
The dogs that didn’t bark

|
“Silver Blaze,” one of the most popular Sherlock Holmes stories written by Sir Arthur Conan Doyle, is a tale about the mysterious kidnapping of a prize race horse on the eve of an important race, and the apparent murder of its trainer. At one point in the story, the Scotland Yard detective, Inspector Gregory, asks Holmes: “Is there any other point to which you would wish to draw my attention?” “To the curious incident of the dog in the night-time,” answers Holmes. Inspector Gregory objects: “The dog did nothing in the night-time.” “That was the curious incident,” remarks Holmes.
This telling exchange inspired The Curious Incident of the Dog in the Night-Time, a 2003 best-selling novel by Mark Haddon.
The reason why I bring this up is that although the January 4 press release, “The origins of abiotic life,” was featured in Phys.org and Science Daily, it seems to have attracted remarkably little comment. Professor Jerry Coyne hasn’t mentioned it over at his Website, Why Evolution Is True. Nor has Professor Larry Moran said anything about it on his Sandwalk blog. Professor P.Z. Myers hasn’t said a word about it on his Pharyngula blog, either. This is quite remarkable, considering that three whole days have elapsed since the University of Groningen’s press release.
Why this deafening silence from North America’s leading evolution advocates? The only reason I can think of is that they don’t think that the new paper by Professor Otto and his team has any real relevance to the origin of life on Earth. What does that tell you about the paper?
But it would be churlish of me to dismiss a paper simply because no-one else had bothered to comment on it. So without further ado, I’d like to enumerate seven reasons why I think the paper fails to shed light on the origin of life on Earth, and of new species.
Seven reasons why the new paper fails to shed light on the origin of life and of new species
1. The peptide structures were the product of intelligent design
The Methods section of the paper (by Sandownik, Mattia, Nowak and Otto) describes how the peptide library was prepared: “Peptide building blocks 1 and 2 were synthesized by Cambridge Peptides Ltd from 3,5-bis(tritylthio)benzoic acid, which was prepared via a previously reported procedure…”. Building blocks 1 and 2 consisted of “an aromatic core functionalized with two thiol groups and a peptide chain”. The paper’s authors produced a dynamic combinatorial library from “building blocks that can react with each other through reversible covalent chemistry combine and recombine to give rise to a diverse set of products”.
The paper also states:
“Each building block is equipped with two thiol groups which, when oxidized to disulfides, form macrocyclic species of different ring sizes. The peptide sequence is designed to have alternating hydrophilic and hydrophobic residues to promote self-assembly into parallel β-sheets.”
(The following very brief explanation is intended for the benefit of readers who don’t have a strong background in chemistry. 3,5-Bis(tritylthio)benzoic acid looks like this and has the chemical formula C45H34O2S2. The term “aromatic” may be used to refer informally to any chemicals derived from the hexagonal, ring-shaped hydrocarbon, benzene, although it can also broadly refer to any flat, cyclic molecule that’s highly stable: for instance, the double-ringed bases in RNA and DNA are also described as aromatic. A thiol group is simply an -SH group, where S represents a sulfur atom and H represents a hydrogen atom. A disulfide refers to a functional group with the general structure R–S–S–R: the two sulfur atoms in the middle are bonded to one another, and the two R’s are groups of atoms containing carbon and/or hydrogen. A peptide is a sequence of amino acid molecules which are bonded together in a short chain. Finally, a molecule which is attracted to water is called hydrophilic, while one which is not attracted to water is called hydrophobic.)
An anonymous biochemist whom I contacted has forwarded his comments to me. The following is a brief summary of his remarks:
This is a very well-designed and skillfully performed piece of chemical engineering (based on some very nicely done research), in which peptide building blocks (as distinct from pure natural peptides) were made by a chemist, not via a natural process. Hopwever, its significance in relation to the origin of biological replication is highly questionable.
Additionally:
The description of the study shows how strong the design component was in these experiments. The carefully ordered formation of covalent bonds was guided by the designed structure of the building blocks, and the alternating hydrophobicity and hydrophilicity was designed to promote the formation of higher order structures.
I have to ask: since when does evidence of Intelligent Design count as evidence for unguided evolution?
2. The reactions would never occur under realistic conditions – and if they did, they’d rapidly come to a halt
The biochemist whom I contacted didn’t think that the conditions in the experiment were very realistic, either. He also pointed out that the reactions described in the paper would soon grind to a halt, under natural conditions. The following two paragraphs are intended to convey the gist of his comments.
This experiment is far removed from OOL [origin of life] conditions, where the reactions and structures would occur at random, and where a very large number of “undesirable” reactions would inevitably occur, drastically reducing the chances of obtaining the “correct” reactions under the assumed OOL conditions.
Under natural OOL conditions, a large number of unwanted reactions could (and probably would) occur, in a solution containing a multitude of different chemical components. Instead of obtaining a functioning system with life-like properties, the end result would be chaotic and unpredictable. Additionally, under natural conditions, the chemical reactions could lead to a dead end, and they would probably not be very useful for generating a replication system from nucleic acids. If this kind of self-organization were common in nature, then we could end up with a very large number of competing systems, which would rapidly deplete the chemical raw materials being used to build nucleic acids. In practice, however, hydrolysis and decomposition, as well as the formation of large amounts of “unwanted” chemical products, would seem to be the dominant trends.
Despite these criticisms, the biochemist whom I contacted wished to compliment the authors of the paper, on the quality of their scientific work. He added that it would indeed be possible for intelligent chemists to build systems that were capable of undergoing intelligently guided molecular evolution, as Otto et al. have done, and he expressed his opinion that they had actually generated a very interesting “evolutionary molecular system.”
3. The structures observed could not possibly have been precursors to the first living organisms on Earth
Another reason why the new paper by Sandownik, Mattia, Nowak and Otto fails to shed light on the origin of life on Earth is that the structures which they created are totally unrelated to those found in living things today. Want proof? I would invite readers to have a look at the article, Diversification of self-replicating molecules in Nature Chemistry, and scroll down and click on Figure 1: Library synthesis and the mechanism of self-replication. Have a look at the ring-structures created by the team of researchers. You’ll notice the six-rings and seven-rings described in the Youtube video at the top of this post. That’s what Professor Otto’s team created.
And now have a look at the diagram below, which depicts three possible representations of the three-dimensional structure of the protein triose phosphate isomerase. Hideously complicated, isn’t it? That’s what life is like. I don’t see any nice little rings of six, seven or eight molecules each. Do you?
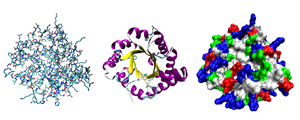
|
I have focused on proteins here, because they’re the next step up from polypeptides. A protein consists of one or more long chains of amino acid units (or residues, in chemical jargon). A protein contains at least one long polypeptide. Short polypeptides, containing less than 20-30 amino acid units (or residues), aren’t usually considered by biochemists to be proteins. Instead, they’re just called peptides, or oligopeptides.
Now my point is that if the peptide structures created by Professor Otto and his team don’t look anything like proteins (much less anyother biological molecule found in living things), then we can safely assume that their relevance to the origin of life on Earth is: nil, nada, nothing, zip, zilch. Zero.
4. There are good reasons to believe that life didn’t begin with a self-replicating molecule
In any case, there are solid scientific reasons for rejecting the idea that life on Earth began with a self-replicating molecule – in which case, the claim by Professor Otto and his team to have created such a molecule is neither here nor there.
The late Professor Robert Shapiro (1935-2011) explained what’s wrong with the replicator-first theory in an interview with Vlad Tarko of Softpedia (Life Did Not Appear with A Self-Replicating Molecule, May 17, 2006):
A scientist proposes an alternative theory to the “replicator” theories of the origin of life – the idea that a self-replicating molecule, such as RNA, has spontaneously appeared and then spread and diversified.
Robert Shapiro from New York University calls such a possibility a “stupendously improbable accident”, although chemists managed to create “prebiotic” syntheses in the lab – syntheses of various building blocks of life such as amino acids. Shapiro says that the use of modern apparatuses and purified reagents is very unlikely to mimic the actual conditions on early Earth.
He says that one of the problems of replicator theories is that a high diversity of molecules of all sorts seems to hamper and endanger the replicator. The mere complexity of the assumed original replicator makes it to be unstable. He argues that what probably happened was the exact opposite – chemical variety was probably beneficial and increased the probability of life. The issue is how this chemical diversity eventually turned into self-replicating chemicals – i.e. life…
The appearance of a molecule that can self-replicate was not the first step, because this requires the combination of diverse chemicals in a long sequence of reactions in a specific order.
Of course, Professor Robert Shapiro’s “metabolism first” theory of the origin of life on Earth faces its own problems, as Leslie Orgel pointed out in an article titled, The Implausibility of Metabolic Cycles on the Prebiotic Earth (PLoS Biology 6(1): e18. doi:10.1371/journal.pbio.0060018 – see here for a non-technical overview by microbiologist Rich Deem). But the point I wish to make is that the “replicator-first” theory espoused by Professor Otto and his team is a very fragile one. It cannot work without just the right sequence of reactions occurring in just the right order, in just the right environment (to ensure that no decomposition occurred, along the way) – in other words, a miracle.
5. The new structures are not alive, in any meaningful sense of the term
Former “Science” editor-in-chief Daniel Koshland Jr. attempted to define “life” in a widely-cited article titled, “Seven pillars of life” (Science 22 March 2002: Vol. 295 no. 5563 pp. 2215-2216, DOI: 10.1126/science.1068489). Koshland listed what he saw as the seven defining features of life:
(1) a program, i.e. “an organized plan that describes both the ingredients themselves and the kinetics of the interactions among ingredients as the living system persists through time” (Koshland, 2002, p. 2215);
(2) improvisation, or a way of changing its master program (achieved on Earth through mutation);
(3) compartmentalization (a surface membrane or skin, and for large organisms, a subdivision into cells, in order to preserve the ingredients required for chemical reactions at their required concentrations);
(4) energy (which on Earth comes from the Sun or the Earth’s internal heat), to keep living systems metabolizing;
(5) regeneration (this includes reproduction), to compensate for the wear and tear on a living system;
(6) behavioral adaptability to environmental hazards; and
(7) seclusion, or some way of preventing one set of chemical reactions from interfering with another, in a cell.
When we look at the peptide structures created by Professor Otto and his team, what do we find? Which conditions are satisfied?
First, the structures lack a master program. Second, lacking a master program, they also lack the ability to modify their master program. Third, the structures possess no internal compartments whatsoever. While they have access to a source of energy (heat), they don’t metabolize, so Koshland’s fourth condition isn’t satisfied, either. (That’s why Professor Otto’s reference to “food” in the press release is misleading.) However, the fifth and sixth conditions are met, in some fashion: the structures can replicate, and they adapt to changes in the availability of different kinds of building blocks. Finally, the structures possess no mechanism for preventing one set of chemical reactions from interfering with another.
Overall Score: 2 out of 7. That’s a pretty long way from what I’d call “life.”
Of course, if you’re intellectually lazy, and you want to define the term “life” to mean anything that can replicate, then don’t let me stop you. But by the same token, many other things would also qualify as alive. As Dr. Steve Wolfram points out in his book, A New Kind of Science (Wolfram Research, 2002, p. 824, it has been known since the 1950s that abstract computational systems possess this capacity as well. Computer viruses would also qualify as alive, on the definition proposed, and there are mechanical devices (such as the “RepRap” machine shown below) that can make copies of themselves, too. How many readers would want to call these devices “alive”?

|
6. The term “mutant” is an inappropriate way of describing the variants that arose
In their press release, Professor Otto and his team use the term “mutant” to refer to a replicating molecule which is slightly different from the original version, because it tends to specialize in different building blocks, when assembling itself. However, as we have seen from Daniel Koshland’s article, “The Seven Pillars of Life“, which I cited above, the word “mutation” properly refers to an organism’s ability to change its master program. Since the structures described in the University of Groningen press release don’t possess anything analogous to a master program, or a genetic code, it follows that they can’t properly be said to mutate.
7. The processes observed in the lab shed no light on speciation, whatsoever
Finally, the processes described in the press release by Professor Otto and his team have absolutely nothing to do with speciation in living things. How can I be so sure of that? There are two things that give the game away.
The first is a very damaging admission in the last paragraph of the press release:
The next step is to introduce death. This can be done by feeding the system a constant flow of building blocks, while draining the contents of the reaction vessel. Replicators can only survive in this system when their growth rate exceeds the removal rate. ‘We could then seed such a system with one set of replicator mutants, and then change the environment, for example by adding another solvent. This would change the fitness of the various replicator mutants and shift the population of mutants towards those that are best at replicating in the new environment.’ The result would be a form of natural selection that Darwin would recognize. ‘We’re not the only ones to be really excited about these experiments – the evolutionary biologist I’ve consulted is too.’
Stop right there. The structures created by Professor Otto and his team don’t die. Consequently, they don’t undergo natural selection. The term, “survival of the fittest,” simply doesn’t apply to them, because they don’t even possess the property of “fitness.”
Without natural selection, speciation which results from a population becoming reproductively isolated from a founder population as it enters and colonizes a new niche, would never get off the ground. Such a model assumes that the new population becomes “fitter,” in relation to its new environment, over the course of time – which means that natural selection has to occur. But as we’ve seen, Professor Otto and his team haven’t achieved natural selection yet. Their excitement is, to say the very least, premature.
The other give-away is that the press release makes no mention whatsoever of a genetic code. And without a genetic code, there can be no genes – and hence, no genetic drift. That rules out other mechanisms of speciation, which rely on the occurrence of genetic drift in a population, in order to achieve reproductive isolation.
No natural selection, no genetic drift: no speciation. Of course, there are also species which are created through hybridization, but that presupposes the existence of genes and sexual reproduction – neither of which are found in the peptide structures created by Professor Otto and his team.
I conclude that the January 4 press release by the University of Groningen on the research conducted by Professor Otto and his team has very little relevance for the origin of life on Earth via an unguided process, much less the origin of living species.
What do readers think?